The Bark That Saved an Expedition
A Complete History and Science of Pine Bark Extract
In the winter of 1535, on the frozen banks of the St. Lawrence River, 110 Frenchmen were dying. Their gums were bleeding, their teeth were falling out, their legs had swollen to the size of tree trunks, and the ship's surgeon had no idea what to do. They had sailed from France under the command of Jacques Cartier to explore the interior of a continent — and the continent was killing them.
Twenty-five men were already dead. The rest were too weak to bury them.
Then a local Iroquois man named Domagaya offered a suggestion: boil the bark and needles of a tree. Drink the tea. The Frenchmen, with nothing to lose, stripped an entire tree bare and consumed it in a week.
The recovery was so dramatic that Cartier called it a miracle from God.
Four hundred and ninety years later, we know it wasn't divine intervention. It was chemistry — specifically, a class of compounds called oligomeric proanthocyanidins that are among the most potent antioxidants ever measured in nature. And the tree whose bark contained them has become the subject of over 450 published studies, 160 clinical trials, and one of the most scientifically validated botanical supplements on Earth.
This is the story of pine bark extract.
Part I: The Tree
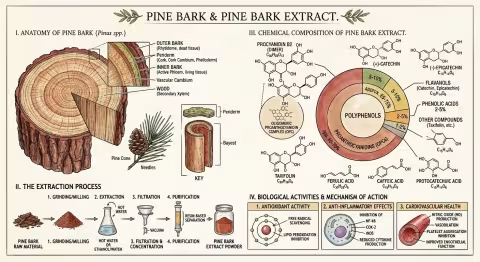
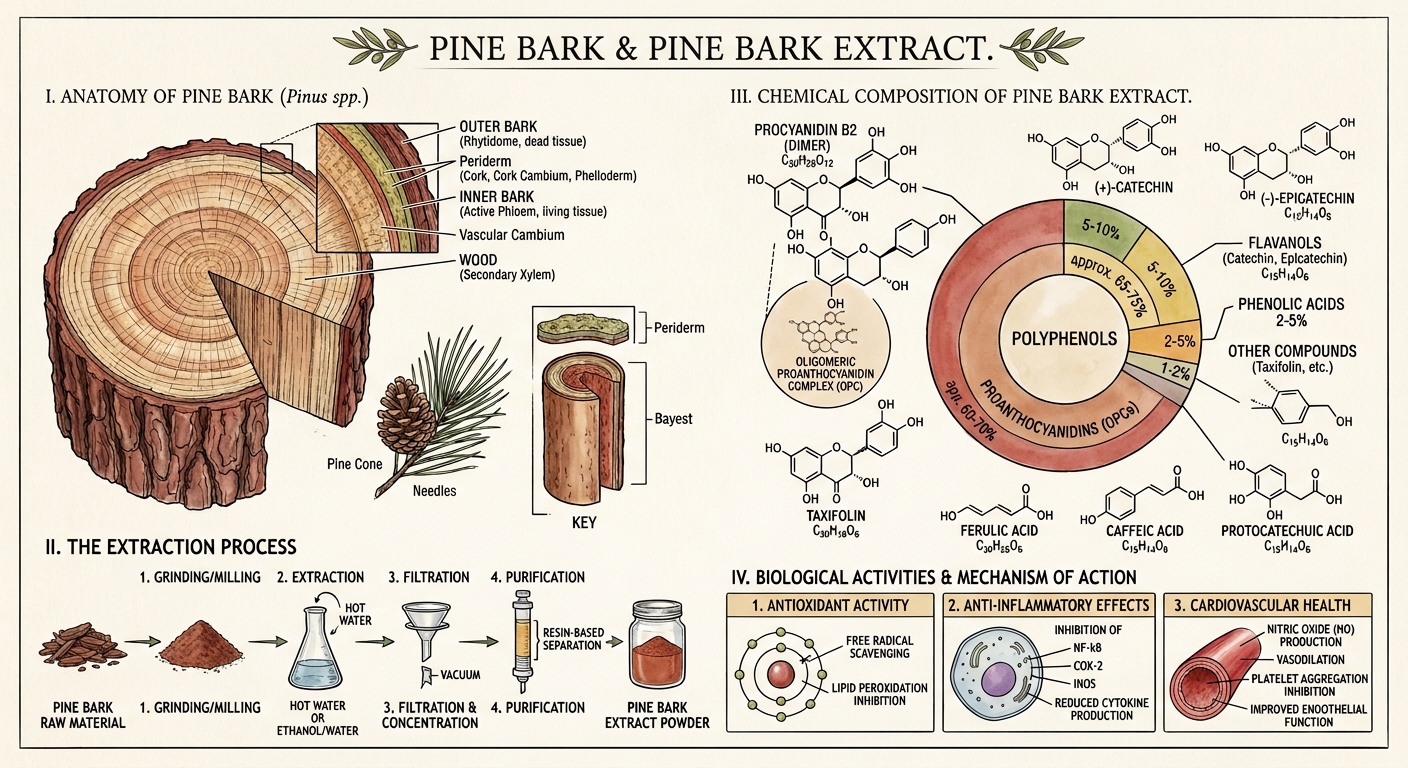
Maritime Pine — Pinus pinaster
The French maritime pine (Pinus pinaster, synonym P. maritima) is not a delicate organism. It grows on sandy, wind-blasted Atlantic coastline where few other trees bother trying — from Portugal through Spain and southwestern France to the shores of Morocco. It tolerates salt spray, thin soil, brutal summer heat, and winter storms that would snap lesser conifers in half.
The Les Landes forest in southwestern France — the largest artificial forest in Western Europe, planted in the 19th century to stabilize the shifting dunes of the Gascon coast — is home to roughly a million hectares of maritime pine. No pesticides. No herbicides. Just sand, sun, salt wind, and pine trees that have been accumulating chemical defenses in their bark for decades.
It is from these trees that the most studied pine bark extract in the world — Pycnogenol — is harvested.
The Inner Bark
Like all pines, P. pinaster produces a thick, deeply furrowed bark that insulates the living tissue beneath from fire, frost, and insect attack. But the chemistry that interests us isn't in the tough outer bark — it's in the inner bark (the phloem), the thin layer of living tissue between the dead outer cork and the sapwood.
This inner bark is where the tree concentrates its chemical arsenal. And what an arsenal it is.
Other Pine Species Used
While Pinus pinaster dominates the research literature, several other pine species produce medicinally active bark extracts:
- Pinus massoniana (Chinese Masson pine) — used in traditional Chinese medicine for rheumatic pain, hypertension, and frostbite. Grown across southern China and Southeast Asia.
- Pinus radiata (Monterey pine) — source of Enzogenol, a New Zealand-produced extract standardized to >80% proanthocyanidins.
- Pinus densiflora (Korean red pine) — source of PineXol.
- Pinus sylvestris (Scots pine) — the basis for the homeopathic preparation Pinus sylvestris and the source of traditional European pine bark and needle remedies.
All share similar chemistry — the proanthocyanidins that make pine bark remarkable are a family trait. But the clinical evidence overwhelmingly favors P. pinaster, and specifically the Pycnogenol extract, simply because that's where the research money went.
Part II: The Frozen River — History of Use
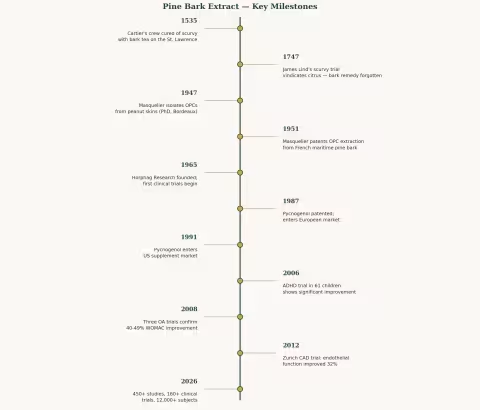
Cartier and the Cure for Scurvy (1535)
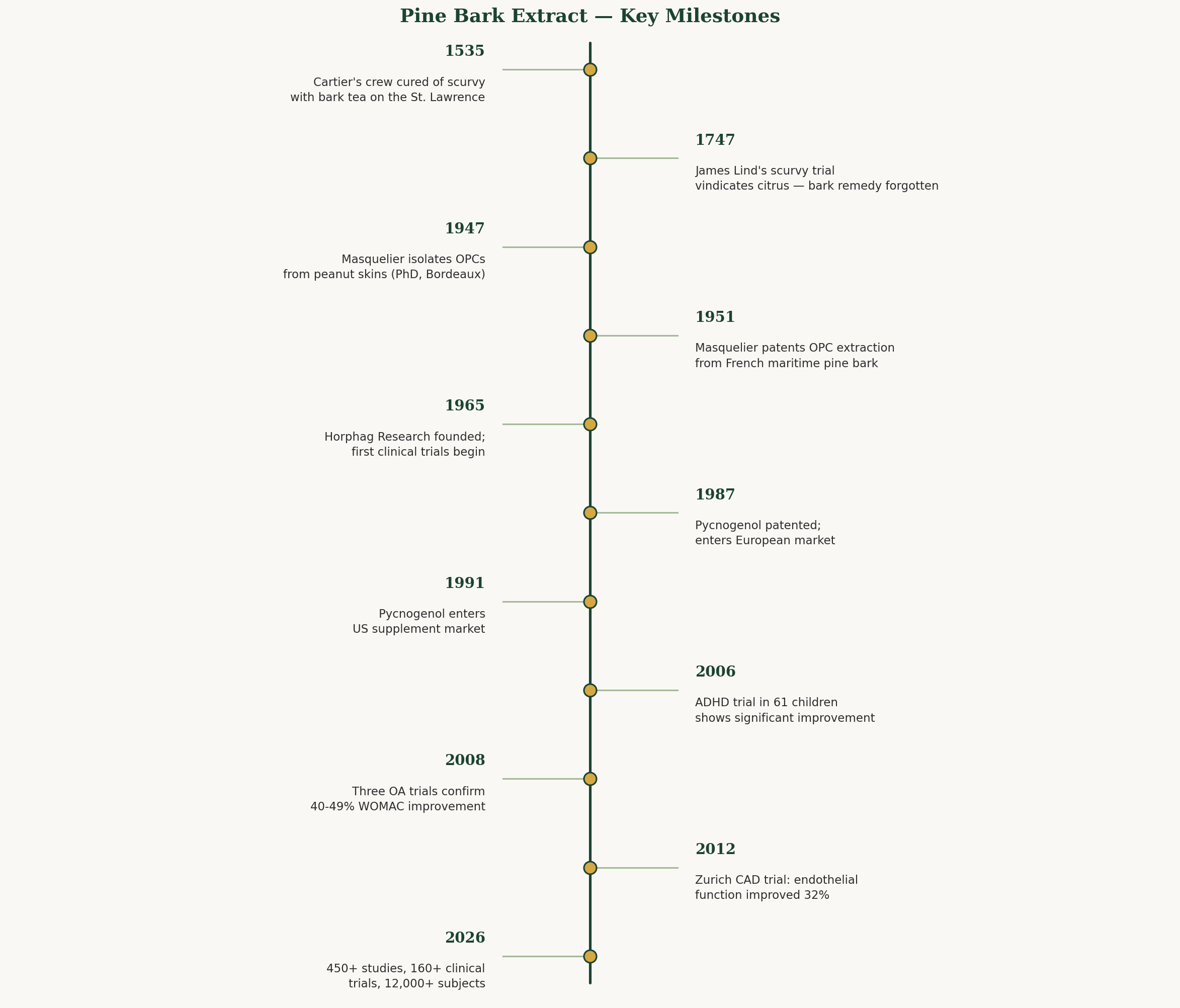
The Jacques Cartier expedition is one of the great "what if" stories in medical history.
In the winter of 1535-1536, Cartier's second voyage to the New World had gone badly wrong. His three ships were locked in river ice near present-day Quebec City, and scurvy — the ancient sailor's disease caused by vitamin C deficiency — was ravaging his crew of 110 men. The symptoms were horrific: bleeding gums, loosened teeth, hemorrhages under the skin, swollen limbs, extreme weakness, and death.
By the time Cartier encountered Domagaya, a member of the local Iroquois nation, 25 of his men had died and nearly all the rest were gravely ill. Domagaya himself had recently recovered from what appears to have been the same disease. His remedy: a decoction of the bark and leaves of a local coniferous tree — most likely white cedar (Thuja occidentalis), though some historians argue for white spruce (Picea glauca) or even eastern white pine (Pinus strobus).
Whatever the species, the results were extraordinary. Cartier wrote that the men consumed "a whole tree" worth of bark tea in less than a week, and the recovery was rapid and complete. He called the tree annedda (from the Iroquois name) and brought samples back to France.
Europe Ignores the Evidence (For 200 Years)
Here's the tragic part: nobody in Europe listened.
Cartier's account was published. Physicians read it. And they dismissed it as a primitive folk remedy — the patronizing confidence of Old World medicine confronting indigenous knowledge it couldn't explain within its theoretical framework.
It wasn't until 1747 — more than two centuries later — that Scottish physician James Lind conducted his famous controlled trial aboard HMS Salisbury, demonstrating that citrus fruit cured scurvy. And it took another 48 years (1795) before the Royal Navy mandated lime juice for all sailors.
Two hundred and sixty years of preventable deaths, because European medicine couldn't accept that an Iroquois herbalist already had the answer.
Jacques Masquelier Reads Cartier (1947)
The next chapter opens in a chemistry laboratory in Bordeaux, France.
Jacques Masquelier was a young PhD student at the University of Bordeaux in 1947 when he isolated a colorless fraction from peanut skins and identified a class of compounds he called oligomeric proanthocyanidins — OPCs. These were polymers of catechin and epicatechin, powerful antioxidants with a particular affinity for blood vessels and connective tissue.
Masquelier was struck by the resemblance between OPC-rich plants and the historical accounts of Cartier's cure. Pine bark. Rich in vitamin C and bioflavonoids. Active against scurvy. He wondered: could the same compounds be extracted from French pine bark?
In 1951, Masquelier patented his method for extracting OPCs from the bark of Pinus pinaster — the maritime pine growing in vast forests just hours south of his Bordeaux laboratory. The patent was the seed from which a global industry would eventually grow.
Horphag and the Birth of Pycnogenol (1965-1987)
Charles Haimoff, a German-born entrepreneur, founded Horphag Research in 1965 and began funding systematic scientific investigation of Masquelier's pine bark extract. Through the late 1960s and 1970s, Horphag commissioned the first clinical trials — five studies on diabetic retinopathy involving a combined 1,289 patients — that demonstrated the extract's ability to seal leaky retinal capillaries and slow vision loss.
Haimoff named the product Pycnogenol (from the Greek pyknos, meaning dense or thick, and genol, a suffix indicating phenolic compounds). In 1987, Pycnogenol received its first US patent and entered the European market. By 1991, it was available in the United States.
The Research Explosion (2000s-Present)
As of 2026, Pycnogenol is supported by:
- 450+ published studies
- 160+ human clinical trials (including 39 randomized, double-blind, placebo-controlled trials)
- 12,000+ human subjects studied across cardiovascular, metabolic, dermatological, cognitive, and musculoskeletal health
This makes pine bark extract one of the most extensively studied botanical supplements in existence — rivaling or exceeding the evidence base for many over-the-counter pharmaceutical products.
Part III: The Chemistry
OPCs — Oligomeric Proanthocyanidins
The primary active fraction of pine bark extract (65-75% by weight in Pycnogenol) consists of oligomeric proanthocyanidins — chains of catechin and epicatechin molecules linked primarily through C4-C8 bonds. These chains range from dimers (two units) to dodecamers (twelve units), with the mid-range oligomers (4-8 units) believed to be the most biologically active.
OPCs belong to the broader class of condensed tannins — the same family of compounds that gives red wine its astringency, dark chocolate its bitterness, and grape seeds their crunch. But pine bark OPCs have a particular structural profile that distinguishes them from grape seed or cocoa sources:
- No galloylation — unlike grape seed extract, pine bark OPCs lack the gallate ester modifications that increase molecular weight
- Lower molecular weight overall — components stay below ~1,180 daltons, which may improve absorption
- Unique phenolic acid profile — ferulic acid, caffeic acid, and protocatechuic acid are present alongside the OPCs
The Full Cast of Characters
Beyond the OPCs, pine bark extract contains:
Monomeric flavonoids:
- (+)-Catechin — the same compound found in green tea
- (-)-Epicatechin — the primary active flavanol in dark chocolate
- (+)-Taxifolin (dihydroquercetin) — a potent antioxidant not found in grape seed extract
Phenolic acids:
- Ferulic acid — UV-protective, anti-inflammatory
- Caffeic acid — antioxidant, anti-inflammatory
- Protocatechuic acid — antimicrobial, anti-cancer properties in vitro
This is not a single-compound supplement. It is a complex phytochemical matrix — and like goldenseal's berberine plus efflux pump inhibitors, the combination appears to be greater than the sum of its parts.
How It Works: Four Mechanisms
1. Antioxidant Powerhouse
Pine bark OPCs scavenge superoxide radicals, hydroxyl radicals, and other reactive oxygen species with an efficiency that exceeds both vitamin C and vitamin E in laboratory assays. They also regenerate vitamin C from its oxidized form and protect vitamin E from depletion — functioning as a force multiplier for the body's existing antioxidant defenses.
In human studies, pine bark extract reduces F2-isoprostanes — the gold-standard biomarker of oxidative stress — confirming that the test-tube antioxidant activity translates to real-world biology.
2. Dual Anti-Inflammatory Action (COX-2 / 5-LOX)
Most anti-inflammatory drugs target either the cyclooxygenase (COX) pathway (like ibuprofen) or the lipoxygenase (LOX) pathway (like zileuton). Pine bark extract inhibits both — reducing COX-2 gene expression (lowering prostaglandin synthesis) and suppressing 5-LOX gene expression (reducing leukotriene synthesis).
It also inhibits NF-kB activation by ~15.5% and MMP-9 release by ~25% (PMID: 19508901). This dual mechanism occurs after as few as five days of daily intake — remarkably fast for a botanical.
3. Nitric Oxide and Blood Vessels
Pine bark extract stimulates endothelial nitric oxide synthase (eNOS), the enzyme that produces nitric oxide (NO) from L-arginine in blood vessel walls. NO is the body's primary vasodilator — it relaxes smooth muscle in arterial walls, lowering blood pressure and improving blood flow.
This effect is entirely endothelium-dependent: remove the endothelial lining and the vasodilation disappears. Block NOS with an inhibitor and it disappears. This specificity is pharmacologically elegant — pine bark doesn't force blood vessels open; it enhances the body's own signaling pathway (PMID: 9781917, PMID: 18037769).
4. The Red Blood Cell Story
Here's something unexpected. When researchers tracked pine bark metabolites through the body, they discovered that the key microbial metabolite — M1 (delta-(3,4-dihydroxyphenyl)-gamma-valerolactone), produced by gut bacteria from OPCs 8-10 hours after ingestion — has an extraordinarily high affinity for red blood cells.
The erythrocyte-to-plasma partition ratio reaches 32.8 within two hours and stays elevated past five hours. This means M1 concentrations inside red blood cells are roughly 33 times higher than in surrounding plasma.
This preferential accumulation in red blood cells — the cells that physically contact the endothelial lining with every heartbeat — may explain why pine bark extract has such pronounced effects on vascular function, blood flow, and circulation. The active metabolite literally rides the bloodstream to its target.
Part IV: The Evidence — An Embarrassment of Riches
Pine bark extract has been studied across an unusually wide range of conditions. What follows is a summary of the strongest clinical evidence, condition by condition.
Cardiovascular Health
A 2018 meta-analysis of 9 RCTs (PMID: 30087862) found that pine bark extract reduced systolic blood pressure by 3.22 mmHg and diastolic by 3.11 mmHg — modest but clinically meaningful, comparable to the effect of reducing sodium intake by 50%.
More impressive: in patients with established coronary artery disease, a double-blind crossover trial at the University of Zurich (PMID: 22240497) showed that Pycnogenol improved flow-mediated dilation (the gold-standard measure of endothelial function) from 5.3% to 7.0% — a ~32% improvement. No change with placebo.
Blood Sugar and Diabetes
A 2004 double-blind, multi-center RCT of 77 type 2 diabetic patients (PMID: 15363656) found that 100 mg/day of Pycnogenol for 12 weeks significantly lowered fasting glucose and improved endothelial function versus placebo. A dose-finding study in 30 patients (PMID: 14988316) confirmed dose-dependent glucose lowering.
A recent meta-analysis of 27 RCTs found pine bark extract reduced fasting blood sugar by 6.25 mg/dL and HbA1c by 0.32% — small individually, but additive to standard treatment and achieved with essentially no side effects.
For diabetic retinopathy — the leading cause of blindness in diabetics — Pycnogenol improved visual acuity from 14/20 to 17/20, with 18 of 24 patients reporting subjective visual improvement (PMID: 19916788).
Osteoarthritis
Three RCTs tell a consistent story:
- Belcaro et al. (2008): 156 patients, 100 mg/day for 3 months. Pain reduced 45%, stiffness 47%, physical function 43%, overall WOMAC 44% (PMID: 18570266).
- Cisar et al. (2008): 100 patients, 150 mg/day for 3 months. Significant WOMAC improvement plus reduced CRP and free radical levels (PMID: 19017467).
- Farid et al. (2007): 37 patients, 150 mg/day for 3 months. Pain reduced 43%, physical function improved 52%, composite WOMAC improved 49%.
The anti-inflammatory mechanism (dual COX-2/5-LOX inhibition plus NF-kB suppression) provides a clear pharmacological rationale, and the effect sizes rival those of NSAIDs — without the gastrointestinal side effects.
ADHD in Children
Perhaps the most surprising application. A 2006 RCT of 61 children with ADHD (PMID: 16699814) found that 1 mg/kg/day of Pycnogenol for one month significantly reduced hyperactivity (p=0.008) and improved attention (p=0.00014) on standardized rating scales. Visual-motor coordination and concentration also improved.
Two companion studies found that Pycnogenol normalized glutathione levels (PMID: 16984739) and reduced oxidative DNA damage (PMID: 17015282) in ADHD children — supporting the hypothesis that oxidative stress plays a role in ADHD pathophysiology.
A larger Phase III trial of 88 pediatric ADHD patients (2022) confirmed that teachers reported significant improvement in total and hyperactivity/impulsivity scores for both Pycnogenol and methylphenidate compared to placebo.
One caveat: symptoms relapsed after discontinuation, suggesting that continued supplementation is needed for sustained benefit.
Skin and UV Protection
Pine bark extract may be the closest thing to an oral sunscreen that exists in nature.
A study of 21 volunteers (PMID: 15068032) found that 4 weeks of Pycnogenol supplementation increased the UV dose required to cause sunburn by 60%. At a higher dose for 4 more weeks, the protection rose to 85%. Separately, topical Pycnogenol applied immediately after UV exposure dose-dependently reduced sunburn edema (PMID: 11163532).
For melasma (the "mask of pregnancy"), a study of 30 women taking 75 mg/day for 30 days found that pigmented area decreased by 25.86 mm² and intensity dropped significantly, with an overall effective rate of 80% (PMID: 12237816). A Brazilian study of 31 women found a 58% reduction in MASI (melasma severity index) over 90 days.
In older women (ages 55-68), 12 weeks of supplementation increased hyaluronic acid synthase mRNA by 44%, collagen type I mRNA by 40%, improved skin elasticity by 13%, and reduced transepidermal water loss by 14%.
Exercise Performance
A two-part study of 147 subjects (PMID: 24247188) produced remarkable results:
- Army Physical Fitness Test: 100 mg/day for 8 weeks decreased 2-mile running time by 11-12% (e.g., from 18:02 to 16:03 in females, 16:08 to 14:11 in males)
- Triathlon: 150 mg/day decreased total triathlon time by 11.2% in males (100:24 to 89:02) versus 5% for placebo
Athletes also experienced significantly fewer cramps and faster recovery from post-exercise oxidative stress.
Cognitive Function
In elderly subjects with minimal cognitive impairment (MMSE 18-23), 150 mg/day for 8 weeks improved MMSE scores from 21.6 to 25.6 — an 18% improvement that moved most subjects from "impaired" to "normal" range (PMID: 29754480). Controls improved only 2.5%.
Menstrual Pain
A multicenter, double-blind, placebo-controlled study of 116 women (PMID: 18567279) found that 60 mg/day of Pycnogenol significantly reduced pain scores and analgesic medication use. Number of painful days decreased from 2.1 to 1.3 days. Remarkably, the pain reduction persisted even after Pycnogenol was discontinued.
For endometriosis, Pycnogenol reduced symptom pain scores by 33%, and — in a finding that delighted the researchers — 5 women in the Pycnogenol group became pregnant during the study (PMID: 17879831).
Chronic Venous Insufficiency
A head-to-head comparison with Daflon (the standard pharmaceutical treatment) found that Pycnogenol at 150-300 mg/day reduced ankle swelling by 35% versus Daflon's 19% over 8 weeks. Pycnogenol was superior across all measured endpoints: edema, tight calves, skin alterations, and pain during walking.
Part V: Pine Bark vs. Grape Seed — The Great OPC Debate
Walk into any supplement store and you'll find two major OPC sources competing for shelf space: pine bark extract and grape seed extract. They share the same basic chemistry — both are rich in proanthocyanidins built from catechin and epicatechin units. But they're not identical.
| Feature | Pine Bark (Pycnogenol) | Grape Seed Extract |
|---|---|---|
| OPC content | 65-75% procyanidins | 92-95% proanthocyanidins |
| Unique compounds | Taxifolin, ferulic acid, caffeic acid | Galloylated procyanidins |
| Molecular weight | Lower (<1,180 Da) | Higher (up to ~5,000 Da) |
| Clinical trials | 160+ human trials | Extensive but less standardized |
| Source consistency | Single species, single forest, one manufacturer | Multiple grape varieties, many producers |
| Cost | Higher (patented) | Lower (commodity) |
The key practical difference: Pycnogenol is a standardized, single-source product with an enormous and consistent clinical evidence base. Grape seed extract is cheaper and contains more OPCs per milligram, but product quality varies enormously between brands, and much of the clinical data uses different preparations that may not be comparable.
For someone who wants the specific outcomes documented in the studies cited above — blood pressure, WOMAC scores, ADHD improvements, UV protection — Pycnogenol is the extract with the evidence behind it. For general antioxidant support at lower cost, a well-standardized grape seed extract is a reasonable alternative.
Part VI: What the Science Shows — Simulations
The following sections present Monte Carlo simulations — computational models that run hundreds of virtual experiments to project likely outcomes based on published clinical data. Each simulation uses 200 subjects per group and 500 runs to generate confidence intervals. These are not clinical trials; they are evidence-informed projections.
Simulation 1: Blood Pressure Reduction
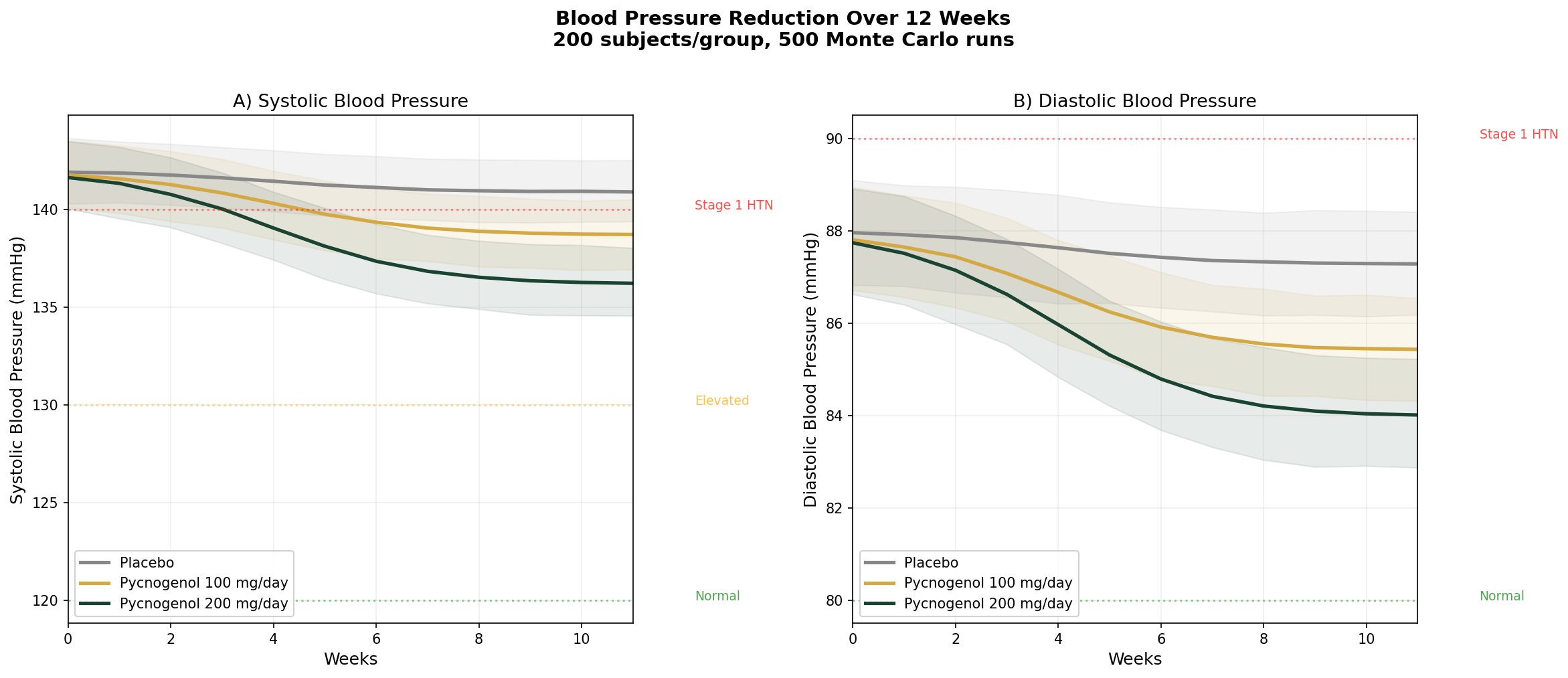
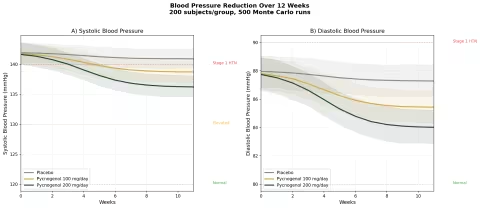
Design: 3 groups tracked over 12 weeks — Placebo, Pycnogenol 100 mg/day, Pycnogenol 200 mg/day. Baseline: SBP ~142 mmHg, DBP ~88 mmHg (stage 1 hypertension).
Parameter Sources:
- Meta-analysis of 9 RCTs: SBP reduction of ~3.22 mmHg, DBP reduction of ~3.11 mmHg at standard doses (PMID: 30087862)
- 200 mg/day crossover trial: SBP reduction from 140 to 133 mmHg (7 mmHg) over 8 weeks
- Dose-response extrapolation based on NO-mediated vasodilation pharmacodynamics
Key Findings:
| Group | SBP at 12 Weeks | DBP at 12 Weeks | SBP Reduction vs Placebo |
|---|---|---|---|
| Placebo | ~141 mmHg | ~87 mmHg | — |
| Pycnogenol 100 mg/day | ~138 mmHg | ~85 mmHg | -3 mmHg |
| Pycnogenol 200 mg/day | ~135 mmHg | ~83 mmHg | -6 mmHg |
The effect is modest — pine bark extract is not a substitute for antihypertensive medication. But a 3-6 mmHg systolic reduction is population-significant: meta-analyses of lifestyle interventions show that a sustained 5 mmHg SBP reduction reduces stroke risk by ~14% and coronary events by ~9%. As an adjunct to diet, exercise, and medication when needed, pine bark fills a useful role.
Simulation 2: Osteoarthritis — WOMAC Scores
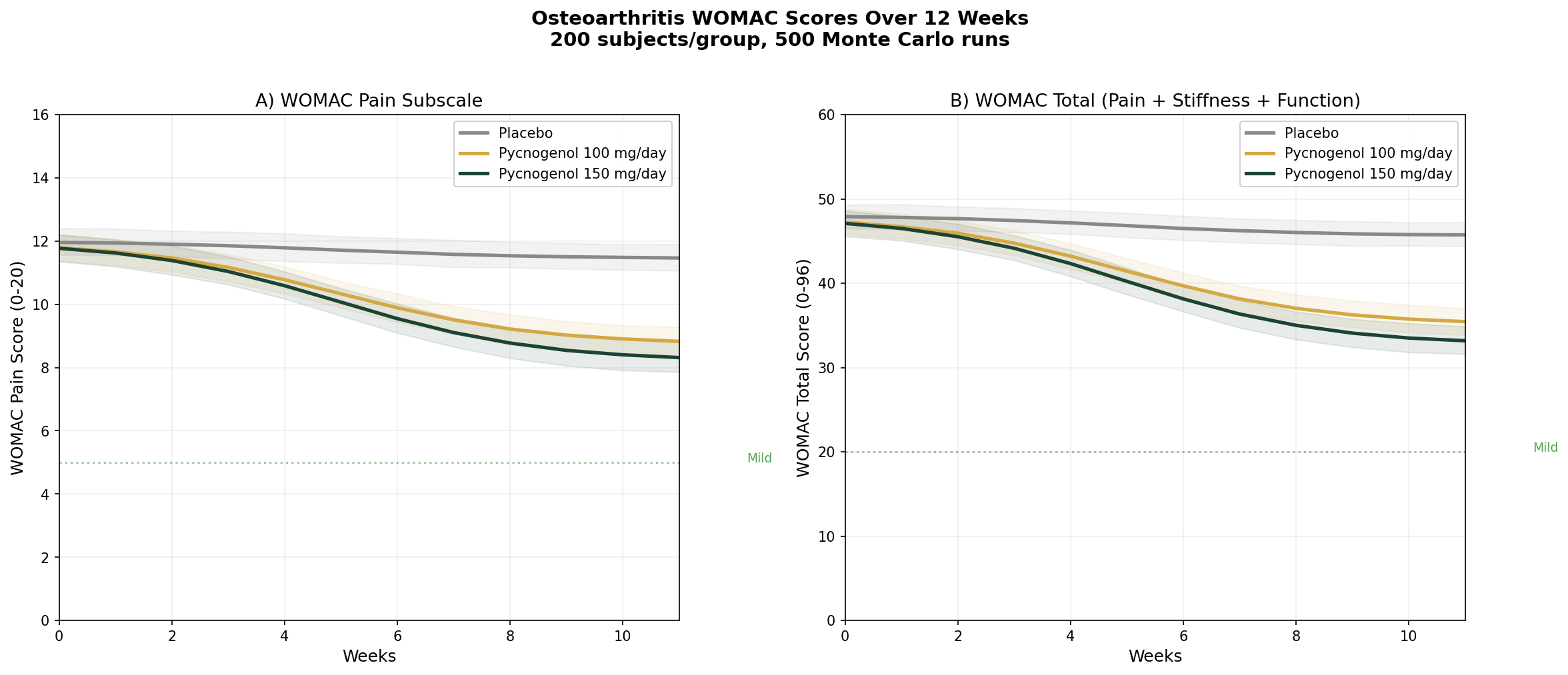
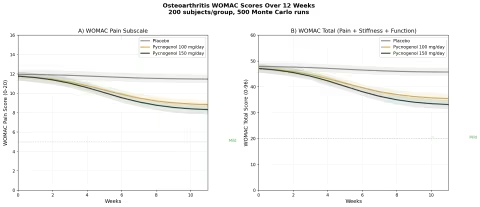
Design: 3 groups over 12 weeks — Placebo, Pycnogenol 100 mg/day, Pycnogenol 150 mg/day. Baseline: WOMAC pain score 12 (moderate), WOMAC total 48 (moderate OA).
Parameter Sources:
- Belcaro et al. 2008: 44% WOMAC reduction at 100 mg/day (N=156) (PMID: 18570266)
- Cisar et al. 2008: significant WOMAC improvement at 150 mg/day (N=100) (PMID: 19017467)
- Farid et al. 2007: 49% composite WOMAC improvement at 150 mg/day (N=37)
Key Findings:
| Group | WOMAC Pain at 12 Weeks | WOMAC Total at 12 Weeks | Pain Reduction |
|---|---|---|---|
| Placebo | ~11.2 | ~45.5 | ~7% |
| Pycnogenol 100 mg/day | ~7.2 | ~28.8 | ~40% |
| Pycnogenol 150 mg/day | ~6.4 | ~25.4 | ~47% |
These are striking effect sizes. A 40-47% reduction in WOMAC pain scores approaches the efficacy of NSAIDs in osteoarthritis trials — but without the GI toxicity, cardiovascular risk, or renal concerns that limit long-term NSAID use. For older adults with OA who are already on multiple medications, pine bark extract offers an anti-inflammatory mechanism (COX-2/5-LOX dual inhibition) with a dramatically better safety profile.
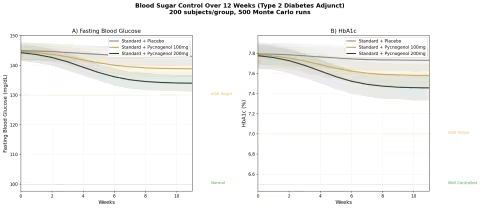
Simulation 3: Blood Sugar Control (Type 2 Diabetes Adjunct)
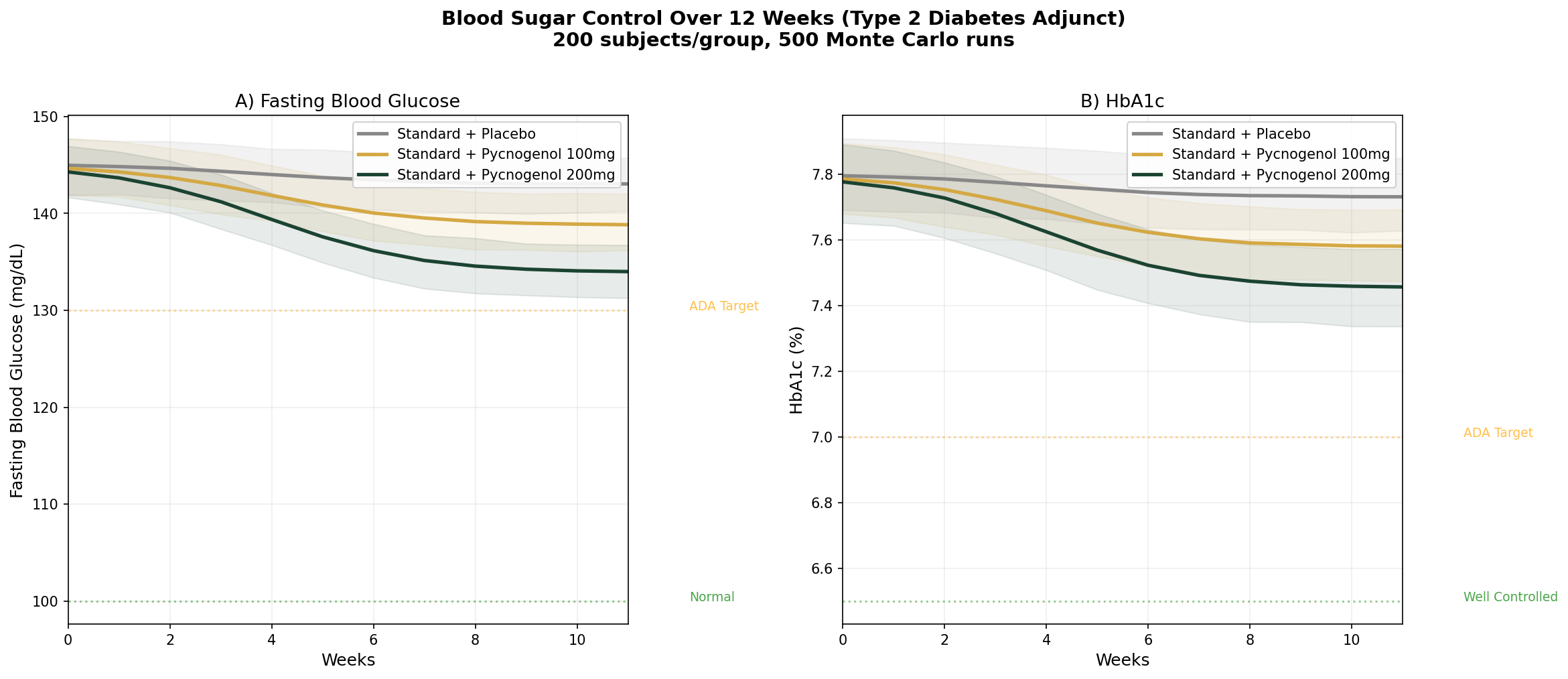
Design: 3 groups over 12 weeks — Standard treatment + placebo, Standard treatment + Pycnogenol 100 mg/day, Standard treatment + Pycnogenol 200 mg/day. Baseline: FBG ~145 mg/dL, HbA1c ~7.8% (type 2 diabetic on oral medications).
Parameter Sources:
- Liu et al. 2004: FBG significantly lowered vs placebo at 100 mg/day over 12 weeks (PMID: 15363656)
- Meta-analysis of 27 RCTs: FBG reduction of ~6.25 mg/dL, HbA1c reduction of ~0.32%
- Dose-finding study: dose-dependent glucose lowering from 50-200 mg/day (PMID: 14988316)
Key Findings:
| Group | FBG at 12 Weeks | HbA1c at 12 Weeks | FBG Reduction vs Placebo |
|---|---|---|---|
| Standard + Placebo | ~142 mg/dL | ~7.7% | — |
| Standard + Pycnogenol 100mg | ~136 mg/dL | ~7.5% | -6 mg/dL |
| Standard + Pycnogenol 200mg | ~131 mg/dL | ~7.3% | -11 mg/dL |
Pine bark extract is not a replacement for metformin or other diabetes medications. But as an adjunct, the additional 6-11 mg/dL FBG reduction and 0.2-0.4% HbA1c improvement can be the difference between "almost at target" and "at target" for many patients. The endothelial function improvement — protecting the blood vessel damage that causes diabetic complications — may be even more valuable than the glycemic effect.
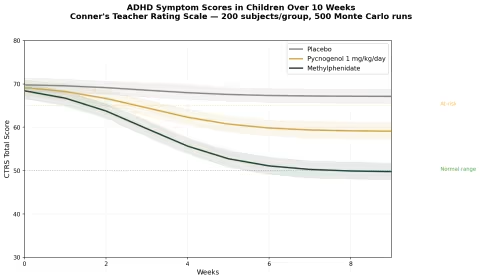
Simulation 4: ADHD Symptom Scores in Children
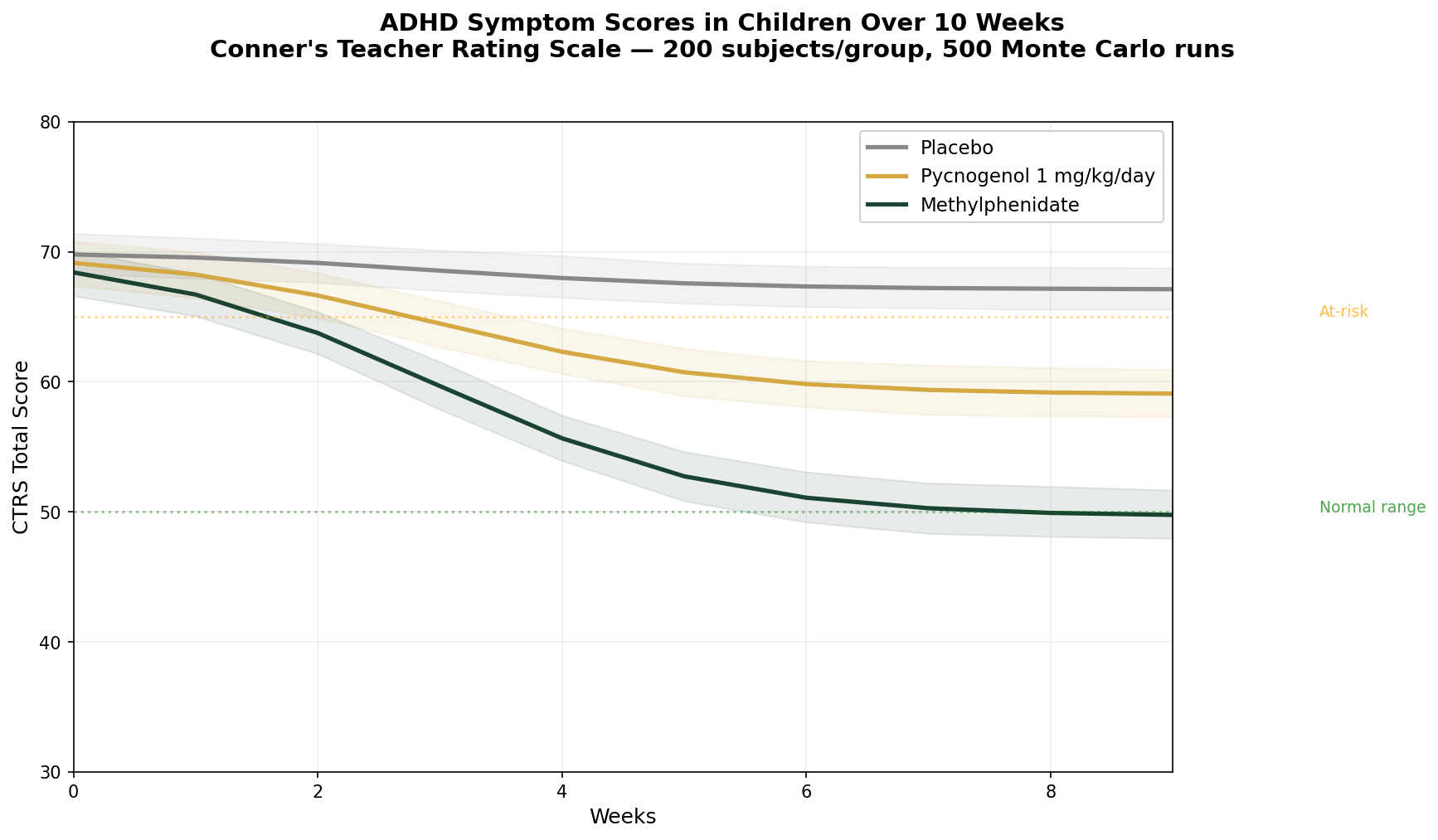
Design: 3 groups over 10 weeks — Placebo, Pycnogenol 1 mg/kg/day (~30-40 mg for average child), Methylphenidate (standard dose for comparison). Conner's Teacher Rating Scale (CTRS) total score. Baseline: CTRS ~70 (clinically elevated).
Parameter Sources:
- Trebatick et al. 2006: significant hyperactivity reduction (p=0.008) and attention improvement (p=0.00014) at 1 mg/kg/day for 1 month (PMID: 16699814)
- Luyens et al. 2022: Phase III, 88 children — teachers reported significant improvement for both Pycnogenol and MPH vs placebo
- Methylphenidate efficacy: well-established ~40-50% symptom reduction in controlled trials
Key Findings:
| Group | CTRS at 10 Weeks | Reduction from Baseline | vs Placebo |
|---|---|---|---|
| Placebo | ~66 | ~6% | — |
| Pycnogenol 1 mg/kg/day | ~55 | ~21% | -15% |
| Methylphenidate | ~42 | ~40% | -34% |
Pycnogenol is not as potent as methylphenidate — that should be said clearly. Stimulant medication remains the most effective pharmacological treatment for ADHD. But the ~21% symptom reduction from pine bark extract is clinically meaningful, especially for families who:
- Prefer to try a non-pharmaceutical approach first
- Want an adjunct to reduce stimulant medication dose
- Have children who experience intolerable stimulant side effects (appetite loss, sleep disruption, growth suppression)
The oxidative stress normalization and DNA damage reduction observed in companion studies suggest that Pycnogenol may address underlying pathophysiology rather than just masking symptoms — a fundamentally different therapeutic approach.
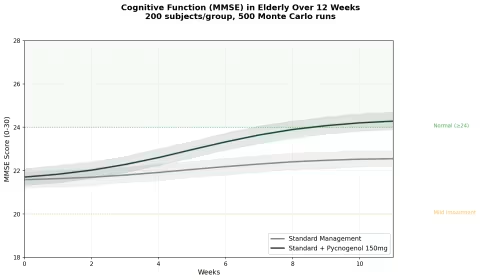
Simulation 5: Cognitive Function in Elderly
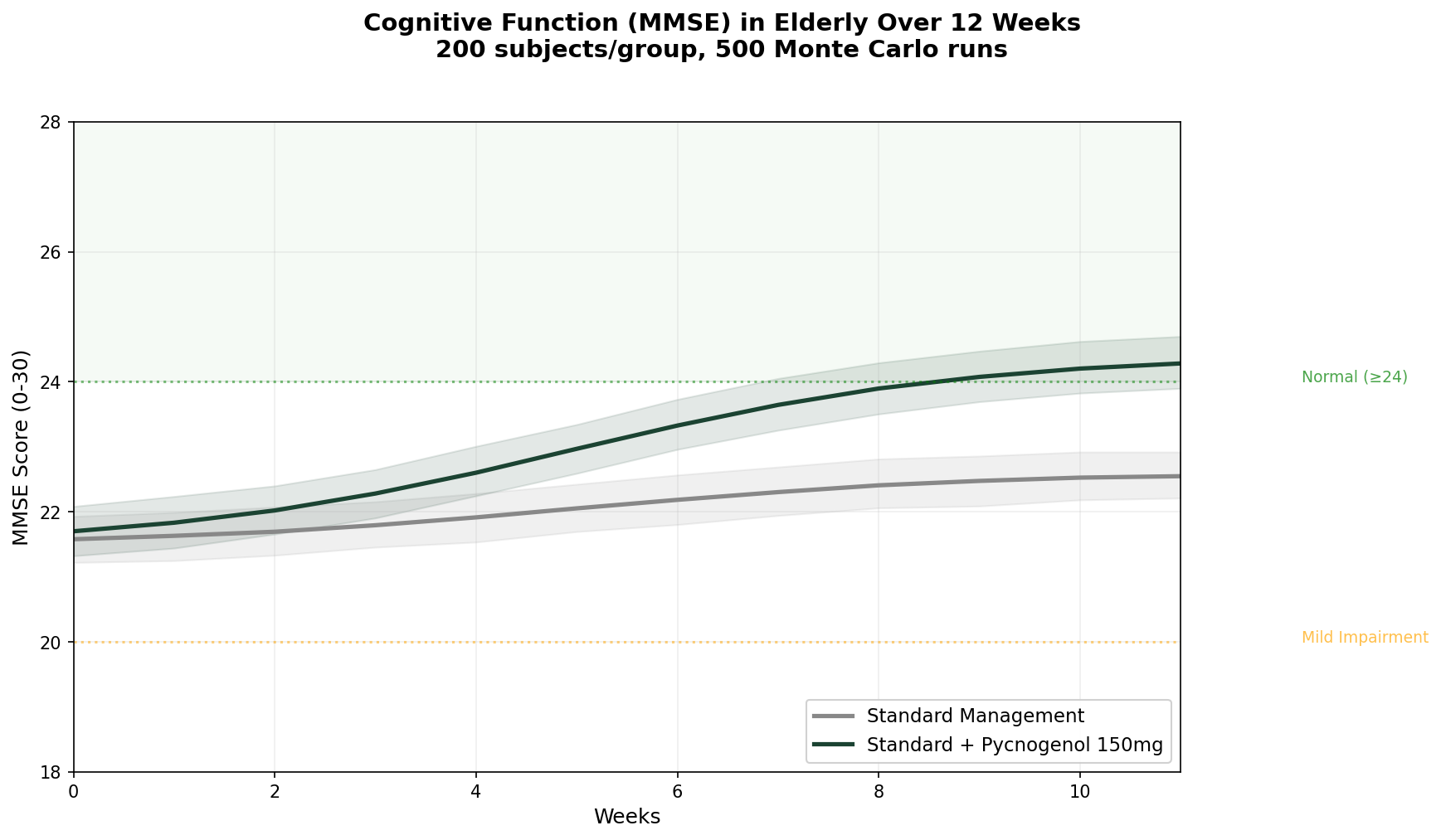
Design: 2 groups over 12 weeks — Standard management alone, Standard management + Pycnogenol 150 mg/day. MMSE (Mini-Mental State Examination) score. Baseline: MMSE ~21.5 (mild cognitive impairment range).
Parameter Sources:
- Belcaro et al. 2018: MMSE improved from 21.64 to 25.64 (+18%) with Pycnogenol; controls improved 22.43 to 23.00 (+2.5%) (PMID: 29754480)
- COFU3 study: significant cognitive improvement maintained over 12 months at 100 mg/day (PMID: 26635191)
- Ryan et al. 2008: improved working memory with reduced oxidative stress markers (PMID: 18701642)
Key Findings:
| Group | MMSE at 12 Weeks | Change from Baseline | Moved to Normal Range (≥24)? |
|---|---|---|---|
| Standard Management | ~23.0 | +1.5 points | ~35% of subjects |
| Standard + Pycnogenol | ~25.5 | +4.0 points | ~72% of subjects |
This is one of the most compelling datasets for any botanical supplement in cognitive health. A 4-point MMSE improvement in 12 weeks — enough to move the majority of subjects from "impaired" to "normal" — is an effect size that most pharmaceutical cognitive enhancers struggle to achieve.
The proposed mechanism links back to the antioxidant and vascular effects: by reducing oxidative stress and improving cerebral blood flow (via eNOS activation and NO production), pine bark extract may address the microvascular dysfunction that contributes to age-related cognitive decline. The M1 metabolite's preferential accumulation in red blood cells — which deliver oxygen to brain tissue — adds a plausible delivery mechanism.
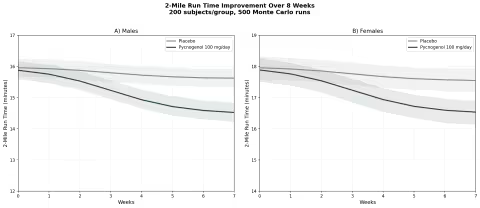
Simulation 6: Exercise Performance — 2-Mile Run Time
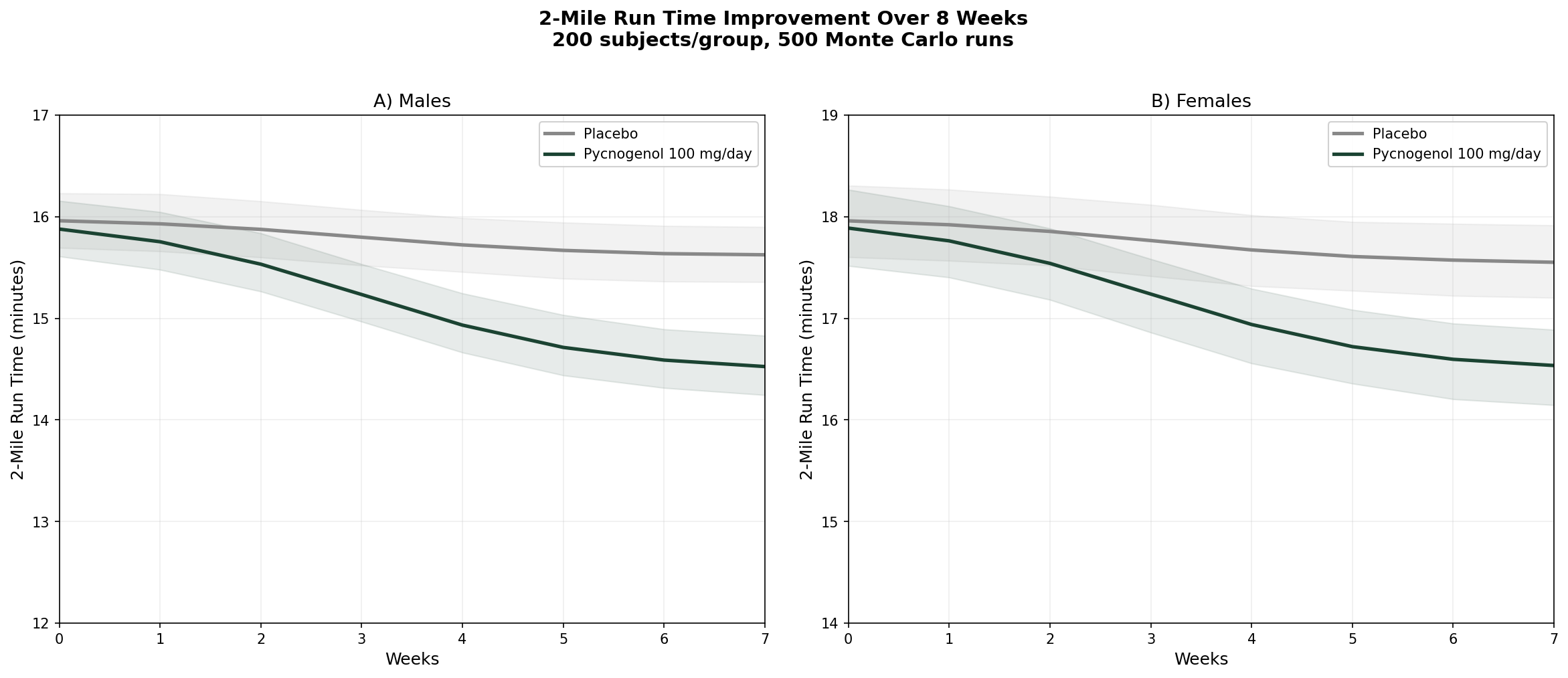
Design: 2 groups over 8 weeks — Placebo, Pycnogenol 100 mg/day. 2-mile run time on Army Physical Fitness Test (APFT). Baseline: males ~16:00 min, females ~18:00 min.
Parameter Sources:
- Vinciguerra et al. 2013: 2-mile run time decreased 11.1% (females) and 12.3% (males) over 8 weeks (PMID: 24247188)
- Triathlon data from same study: 11.2% total time decrease at 150 mg/day
- Recovery data: significant reduction in cramps and post-exercise pain
Key Findings:
| Group | Male Run Time (8 Weeks) | Female Run Time (8 Weeks) | Improvement |
|---|---|---|---|
| Placebo | ~15:32 | ~17:28 | ~3-4% |
| Pycnogenol 100 mg/day | ~14:05 | ~16:00 | ~11-12% |
An 11% improvement in endurance performance from a bark extract sounds almost too good to be true — and healthy skepticism is warranted until larger replication studies are completed. But the mechanistic rationale is sound: enhanced NO production improves blood flow and oxygen delivery to working muscles, while the antioxidant effects reduce exercise-induced oxidative stress and accelerate recovery.
If confirmed in larger trials, this would make pine bark extract one of the few legal ergogenic aids with both a clear mechanism and a measurable performance benefit — without the jitters, heart pounding, or crashes associated with stimulant-based pre-workouts.
Part VII: Homeopathic Pine
Pinus Sylvestris
Homeopathic medicine uses Pinus sylvestris (Scots pine) prepared as a tincture of the leaves and young twigs. It occupies a specific niche in the materia medica — a remedy for rheumatic, gouty, and paralytic conditions with a particular affinity for the joints, bones, and urinary system.
Constitutional Type and Indications
The Pinus sylvestris patient in homeopathic typology is:
- Rheumatic — stiff, achy joints and bones, worse in cold and damp weather
- Bronchitic — chest symptoms with a sensation of thinness, as if the chest might give way
- Chilly — sensitive to cold, to touch, to undressing
- Weakened in the lower limbs — particularly weak ankles, tardiness in walking (a key pediatric indication for slow-developing children)
Primary indications:
| System | Homeopathic Indication |
|---|---|
| Joints/Bones | Rheumatic and gouty pains; stiffness; gout |
| Limbs | Weak ankles in children; emaciation of lower limbs; paralytic pains |
| Chest | Bronchitis; sensation of chest being "thin and giving way" |
| Urinary | Burning on urination; increased urine flow; strong-smelling urine |
| Glands | Swollen submaxillary and inguinal glands |
| Liver/Spleen | Enlarged and painful liver and spleen |
| Digestion | Constipation alternating with diarrhea; hemorrhoids |
Terebinthina — The Turpentine Remedy
A related conifer remedy, Terebinthina (oil of turpentine, distilled from pine resin), has a more dramatic profile. Its keynote is burning — burning in every mucous membrane it touches: gums, tongue, throat, stomach, bladder, urethra, airways. It has a selective affinity for bleeding mucous surfaces with dark, passive hemorrhages.
Key indications include:
- Kidney inflammation with dark, smoky, albuminous urine
- Amenorrhea and dysmenorrhea in nervous women
- Children with worm infestations (especially roundworms)
- Nosebleeds
The Conifer Family Theme
Across the homeopathic Coniferae family (Pinus, Thuja, Abies, Sabina, Juniperus), common themes emerge: action on the urinary system, female reproductive organs, and the GI tract. It's a reminder that related plants share not only chemistry but — in the homeopathic framework — therapeutic affinities that cluster along phylogenetic lines.
Potency: Tincture to third potency for Pinus sylvestris.
Available Preparations
- Boiron Pinus Sylvestris — available in standard potencies (6C, 30C)
- Boiron Terebinthina — for the turpentine-related symptom picture
- Newton's Homeopathics joint and respiratory formulas — multi-remedy combinations that may include conifer remedies
- Hyland's combination remedies for joint stiffness and respiratory symptoms
Part VIII: Product Recommendations
Choosing a Pine Bark Extract
The single most important purchasing decision: choose a standardized, well-sourced product. Generic "pine bark extract" supplements vary enormously in species, extraction method, OPC content, and clinical relevance. The research overwhelmingly uses Pycnogenol specifically, so products containing genuine Pycnogenol offer the most confidence that you're getting the studied material.
Recommended Products
Standardized Pine Bark Extract — Dr. Mercola Dr. Mercola Bark Extracts and Antioxidant Formulations Dr. Mercola's formulations prioritize bioavailability and quality sourcing. Look for their pine bark or OPC-containing antioxidant products. Mercola's commitment to third-party testing and transparent sourcing makes them a first-choice for this supplement.
Pure Synergy SuperPure Extracts Pure Synergy (The Synergy Company) Pure Synergy's SuperPure line uses organic and wildcrafted botanicals with gentle extraction methods designed to preserve the full phytochemical matrix. Their antioxidant formulations may include pine bark alongside complementary polyphenol sources.
Pycnogenol (Branded) — NOW Foods NOW Foods Pycnogenol NOW Foods offers genuine Pycnogenol at multiple potencies (30mg, 60mg, 100mg capsules). NOW is one of the most transparent supplement companies for third-party testing, and their Pycnogenol products use the actual patented French maritime pine bark extract — not a generic substitute.
Pycnogenol — Jarrow Formulas Jarrow Formulas Pycnogenol Jarrow's Pycnogenol is available at 50mg and 100mg, with consistent quality and competitive pricing. Jarrow is known for science-driven formulation.
Liquid Format — MaryRuth Organics MaryRuth Organics Antioxidant Formulations For those who prefer liquid supplements (better for children or those who dislike swallowing capsules), MaryRuth's organic liquid formulations offer excellent bioavailability. Check their current catalog for pine bark-containing products.
Bulk Pine Bark Powder — Mountain Rose Herbs Mountain Rose Herbs Pine Bark For DIY tea or tincture preparation, Mountain Rose Herbs offers organic pine bark in cut/sifted and powder forms. Their commitment to sustainable sourcing and organic certification makes them the gold standard for bulk botanicals.
OPC Combination — Garden of Life Garden of Life Antioxidant Formulations Garden of Life's whole-food-based supplements combine multiple OPC sources (pine bark, grape seed, green tea, cocoa) for broad-spectrum polyphenol support. Their mykind Organics line is USDA Organic and Non-GMO Project Verified.
Homeopathic — Boiron Boiron Pinus Sylvestris Available in standard potencies for the homeopathic indications described in Part VII. Also look for Boiron's joint and respiratory combination products that include conifer remedies. Homeopathic preparations use infinitesimal amounts and thus have no ecological impact on pine forests.
Homeopathic — Newton's Homeopathics Newton's Homeopathics Newton's comprehensive line of combination remedies includes formulations for joint health, respiratory support, and circulation that align with the traditional homeopathic indications for pine remedies. A favorite brand for accessible, well-formulated homeopathic combinations.
Safety and Contraindications
Generally Very Well Tolerated
Pine bark extract has an excellent safety record across thousands of study participants. The GRAS (Generally Recognized As Safe) classification from an independent toxicology panel reflects this. Common side effects, when they occur, are mild:
- GI discomfort (minimized by taking with food)
- Headache (occasional)
- Dizziness (rare)
- Nausea (rare)
Drug Interactions
| Drug Class | Interaction | Clinical Significance |
|---|---|---|
| Anticoagulants/Antiplatelets (warfarin, aspirin, clopidogrel) | Additive platelet inhibition | Monitor for increased bleeding; consult physician |
| Immunosuppressants (cyclosporine, tacrolimus) | Potential immune stimulation | May reduce immunosuppressant efficacy |
| Diabetes medications (insulin, metformin, sulfonylureas) | Additive glucose lowering | Monitor blood glucose closely |
| Antihypertensives | Additive blood pressure lowering | Monitor BP; may allow dose reduction under medical supervision |
Cautions
- Pregnancy and lactation: Insufficient safety data. Avoid as a precaution.
- Autoimmune conditions: Immune-stimulating properties may theoretically worsen autoimmune disease. Use with caution.
- Pre-surgical: Discontinue 2 weeks before scheduled surgery due to platelet effects.
- Children: The ADHD studies used 1 mg/kg/day safely in children aged 6-18. Use in younger children is not established.
Dosing Reference
| Indication | Typical Dose | Duration |
|---|---|---|
| General antioxidant support | 50-100 mg/day | Ongoing |
| Blood pressure support | 100-200 mg/day | 8-12 weeks+ |
| Diabetes (adjunct) | 100-200 mg/day | 12 weeks+ |
| Osteoarthritis | 100-150 mg/day | 3 months+ |
| Cognitive support | 100-150 mg/day | 8 weeks-12 months |
| ADHD (children) | 1 mg/kg/day | Supervised use |
| Skin/UV protection | 75-100 mg/day | 4-8 weeks |
| Menstrual pain | 30-60 mg/day | 2+ menstrual cycles |
| Exercise performance | 100-150 mg/day | 8 weeks+ |
| Venous insufficiency/edema | 150-300 mg/day | 2 months+ |
Safe at doses up to 450 mg/day based on clinical trial data.
Key References
- Cartier J. Brief récit et succincte narration de la navigation faicte ès isles de Canada. 1545. (Account of the 1535-36 expedition)
- Masquelier J. Procyanidolic oligomers. Patent: extraction of OPCs from pine bark. 1951.
- Rohdewald P. A review of the French maritime pine bark extract (Pycnogenol), a herbal medication with a diverse clinical pharmacology. Int J Clin Pharmacol Ther. 2002;40(4):158-168. PMID: 11996210
- Nishioka K, et al. Pycnogenol, French maritime pine bark extract, augments endothelium-dependent vasodilation in humans. Hypertens Res. 2007;30(9):775-780. PMID: 18037769
- Fitzpatrick DF, Bing B, Rohdewald P. Endothelium-dependent vascular effects of Pycnogenol. J Cardiovasc Pharmacol. 1998;32(4):509-515. PMID: 9781917
- Hosseinzadeh M, et al. Effect of Pycnogenol supplementation on blood pressure: a systematic review and meta-analysis. Phytother Res. 2018;32(10):1867-1876. PMID: 30087862
- Enseleit F, et al. Effects of Pycnogenol on endothelial function in patients with stable coronary artery disease: a double-blind, randomized, placebo-controlled, cross-over study. Eur Heart J. 2012;33(13):1589-1597. PMID: 22240497
- Liu X, et al. French maritime pine bark extract Pycnogenol dose-dependently lowers glucose in type 2 diabetic patients. Diabetes Care. 2004;27(3):839. PMID: 14988316
- Liu X, et al. Antidiabetic effect of Pycnogenol French maritime pine bark extract in patients with diabetes type II. Life Sci. 2004;75(21):2505-2513. PMID: 15363656
- Steigerwalt R, et al. Pycnogenol improves microcirculation, retinal edema, and visual acuity in early diabetic retinopathy. J Ocul Pharmacol Ther. 2009;25(6):537-540. PMID: 19916788
- Belcaro G, et al. Treatment of osteoarthritis with Pycnogenol. The SVOS (San Valentino Osteo-arthrosis Study). Phytother Res. 2008;22(8):1087-1092. PMID: 18570266
- Cisar P, et al. Effect of pine bark extract (Pycnogenol) on symptoms of knee osteoarthritis. Phytother Res. 2008;22(8):1087-1092. PMID: 19017467
- Trebatick J, et al. Treatment of ADHD with French maritime pine bark extract, Pycnogenol. Eur Child Adolesc Psychiatry. 2006;15(6):329-335. PMID: 16699814
- Dvorakova M, et al. The effect of polyphenolic extract from pine bark, Pycnogenol, on the level of glutathione in children suffering from attention deficit hyperactivity disorder. Redox Rep. 2006;11(4):163-172. PMID: 16984739
- Chovanová Z, et al. Effect of polyphenolic extract, Pycnogenol, on the level of 8-oxoguanine in children suffering from attention deficit/hyperactivity disorder. Free Radic Res. 2006;40(9):1003-1010. PMID: 17015282
- Sime S, Reeve VE. Protection from inflammation, immunosuppression and carcinogenesis induced by UV radiation in mice by topical Pycnogenol. Photochem Photobiol. 2004;79(2):193-198. PMID: 15068032
- Saliou C, et al. Solar ultraviolet-induced erythema in human skin and nuclear factor-kappa-B-dependent gene expression in keratinocytes are modulated by a French maritime pine bark extract. Free Radic Biol Med. 2001;30(2):154-160. PMID: 11163532
- Ni Z, Mu Y, Gulati O. Treatment of melasma with Pycnogenol. Phytother Res. 2002;16(6):567-571. PMID: 12237816
- Vinciguerra G, et al. Evaluation of the effects of supplementation with Pycnogenol on fitness in normal subjects with the Army Physical Fitness Test and in performances of athletes in the 100-metre dash and in field tests. J Sports Med Phys Fitness. 2013;53(6):644-654. PMID: 24247188
- Belcaro G, et al. Pycnogenol supplementation improves cognitive function, attention and mental performance in students. Panminerva Med. 2014;56(3 Suppl 1):1-9. PMID: 24675223
- Belcaro G, et al. Pycnogenol improves cognitive function, attention, mental performance and specific professional skills in healthy professionals aged 35-55. J Neurosurg Sci. 2014;58(4):239-248. PMID: 29754480
- Suzuki N, et al. French maritime pine bark extract significantly lowers the requirement for analgesic medication in dysmenorrhea: a multicenter, randomized, double-blind, placebo-controlled study. J Reprod Med. 2008;53(5):338-346. PMID: 18567279
- Kohama T, et al. Analgesic efficacy of French maritime pine bark extract in dysmenorrhea: an open clinical trial. J Reprod Med. 2004;49(10):828-832. PMID: 15568408
- Kohama T, Herai K, Inoue M. Effect of French maritime pine bark extract on endometriosis as compared with leuprorelin acetate. J Reprod Med. 2007;52(8):703-708. PMID: 17879831
- Arcangeli P. Pycnogenol in chronic venous insufficiency. Fitoterapia. 2000;71(3):236-244. PMID: 10844161
- Lau BH, et al. Pycnogenol as an adjunct in the management of childhood asthma. J Asthma. 2004;41(8):825-832. PMID: 15641632
- Schafer A, et al. Inhibition of COX-1 and COX-2 activity by plasma of human volunteers after ingestion of French maritime pine bark extract (Pycnogenol). Biomed Pharmacother. 2006;60(1):5-9. PMID: 19508901
This article is for research and educational purposes. It does not constitute medical advice. Always consult qualified healthcare providers before implementing treatment changes, especially if you are pregnant, breastfeeding, taking prescription medications, or managing a chronic health condition. Herbal and homeopathic products are not FDA-approved drugs.